Medical Software Development Made Simple, Safe, and Scalable
Build safe, intuitive, and scalable software for medical devices, lab equipment, and healthcare IT. Qt Group offers end-to-end solutions for medical software design and development, with capabilities demonstrated across safety-critical systems and medical UI and UX design.




































Qt Group across the MedTech industry.
- Medical Devices
- Clinical Lab
- Healthcare IT

Medical Devices
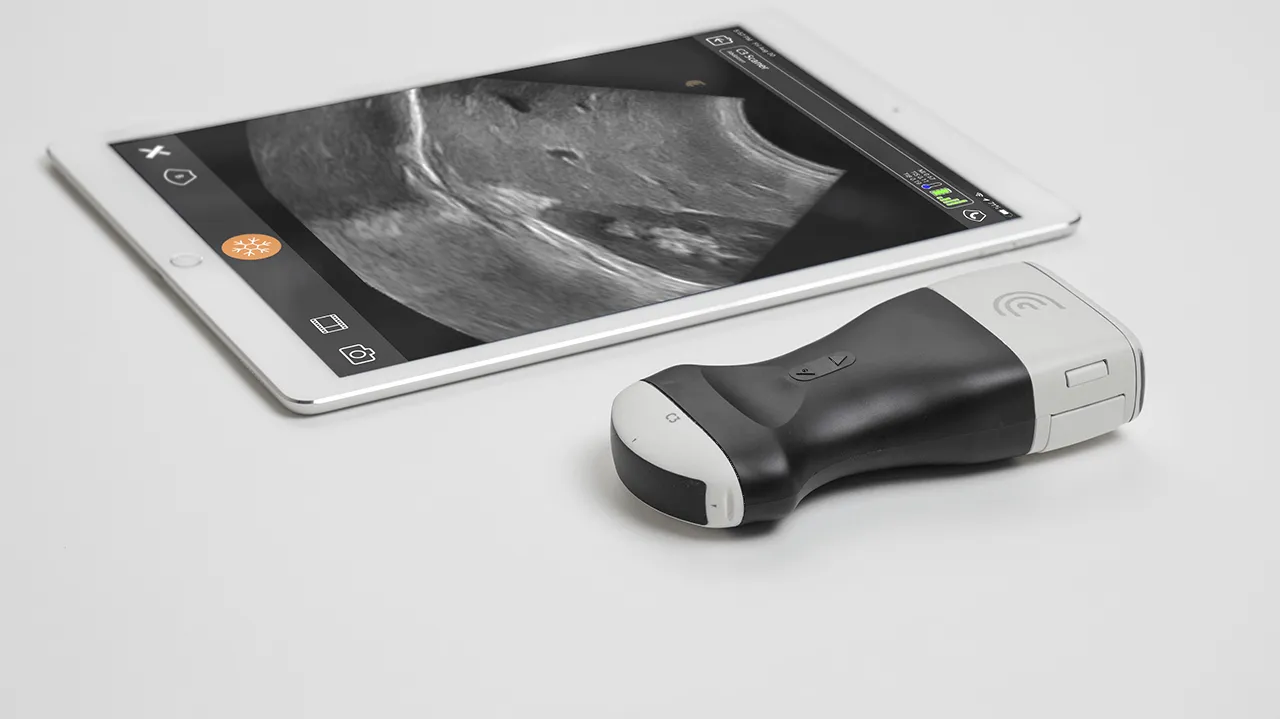
Devices such as patient monitors, robotic surgical systems, and ultrasound devices pose high risks because a malfunction could impact patient safety, and cybersecurity threats compromise device integrity.
With high-quality HMI components, real-time performance, and graphics acceleration, Qt Framework delivers intuitive and responsive medical device user interfaces. Built with cybersecurity in mind, Qt tools further support secure application development. Qt cross-platform capabilities allow for creating and testing applications on a PC or even in the cloud without the need for actual hardware, delivering consistently the best performance across devices. This approach also accelerates time to market by enabling early testing, rapid iteration, and easy deployment.

Clinical Lab
Errors in the lab equipment can endanger patient safety, while cybersecurity breaches expose sensitive patient records. With its rich libraries for high-performance, reliable HMIs, Qt Framework lets clinical lab manufacturers shift focus from low-level tech details to enhanced usability, safety, and efficiency. A clear, intuitive HMI supports personnel on complex tasks ranging from sample preparation and analysis to digital imaging review, data interpretation, and more.
Qt helps medical labs meet the accuracy, automation, and usability requirements essential for lab and diagnostic equipment. The ability to connect seamlessly with laboratory and hospital information systems makes clinical equipment built with Qt fit naturally into the connected lab ecosystem.
Discover More: Safe, Smart, Seamless: Navigating HMI Hurdles in Lab Equipment

Healthcare IT
Healthcare IT systems manage vast sensitive patient data. Breaches can jeopardize patient records, disrupt care, and damage trust. The demand for secure, interoperable, and scalable healthcare IT systems bridging clinicians, patients, and data systems can be easily met with Qt Framework's data processing capabilities, cybersecurity and cyber-resilience support, and networking solutions.
With Qt, Healthcare IT providers can easily create cross-platform applications that run consistently across desktops, mobile devices, and the web, and connect seamlessly to medical devices and clinical labs—simplifying integration with electronic health records, hospital and laboratory information systems, and more.

Efficiently Create Intuitive Medical Software with Ready-Made Components
Reduce time-to-market by accessing and customizing production-proven components and functions already in use in highly regulated industries.
Qt provides easy access to a large set of ready-to-use libraries and customizable UI constructs, including control elements, menus, encryption protocols, secure storage, and DICOM view support, allowing MedTech companies to launch products faster and benefit from long-term support. Companies using Qt gain speed to meet market demands, clarity in development, and confidence that the medical software is safe and compliant.
Qt Group has a strong production history in the medical technology sector with millions of medical devices built with Qt currently on the market that have passed Class I, Class II and Class III FDA certification and Class I, Class IIa, IIb, and III EU certification. Notably, 90% of the top global MedTech companies build their products with Qt, highlighting the sector's trust in Qt for critical healthcare decision-making and patient care.

Verify and Validate Your Medical Software Usability from the Earliest Development Stages
Efficiently drive early clinical and human-factor testing with an integrated design-develop-test workflow enabling rapid error detection and quick iterations.
Clinical lab equipment is used by a range of professionals, from experienced technicians to novices with limited training. Medical UIs have to be intuitive to enable a variety of users to navigate complex workflows easily. Moreover, as healthcare increasingly transitions to home settings and the risk of device misuse rises, regulatory bodies stress the importance of intuitive usability and comprehensive human factor testing in medical device UI design.
Qt Group provides a wide range of products to improve early testing and usability through quick prototypes, real-time user behavior tracking, and automated UI testing. In addition to the possibility of sharing fully functional prototype applications across teams over the web for UI/UX verification (Qt Design Studio), the Qt Group's product family includes automated Quality Assurance tooling for early verification of the software functions (Squish), code coverage (Coco), and compliance with coding guidelines (Axivion).

Meet the Highest Medical Certification Standards
Uphold the highest safety and reliability standards in medical software development by meeting the criteria set by notified bodies.
Whether you’re developing FDA or EU Class II–III medical devices—such as surgical robots, infusion pumps, patient monitoring systems, or medical imaging applications—or clinical lab equipment, Qt helps you balance innovation with compliance. Qt is ISO 9001:2015 certified with an established Quality Management System. Qt Safe Renderer is certified to both IEC 62304 - Medical Device Software, Software Lifecycle Processes, and IEC 61508 - Functional Safety of Electrical/Electronic/Programmable Electronic Safety-related Systems.
Qt Group offers support for global regulatory clearances and certifications, which help improve audit accuracy with consistent information. This is especially relevant for clinical lab equipment that must adhere to regulations such as those from the FDA (U.S.), CE marking (EU), and ISO 13485 standards, which include specific user interface design requirements.
Secure Your Medical Software Against Cyber Threats
Transform your medtech solution into a smart, connected Internet of Medical Things system that is operable from home, clinic, or lab, with secure connectivity and protection against cybersecurity risks.
For Healthcare IT applications like electronic health records, telehealth applications, and remote patient monitors, data protection against cyberthreats is crucial. To comply with standards such as IEC 62304, HIPAA (U.S.), GDPR (EU), HL7, and FHIR, Qt's security-critical code undergoes regular reviews, testing, and documentation. This process adheres to cybersecurity and cyberresilience best practices, including mandatory code reviews, merit-based inspections, continuous integration, and timely security updates. Being maintained for years with security updates, patches, and a stable roadmap, Qt long-term releases secure healthcare product’s permanence on the market over time.
Qt QA tools and third-party assessments, such as penetration testing, help detect vulnerabilities early. Additionally, tools like Qt Application Manager isolate applications and bind them to specific hardware, reducing the risk of external interference and data leakage, and ensuring system integrity across devices, labs, and clinical settings. The tool gives developers full control of applications in the stack to protect sensitive patient data in medtech environments.
Decentralized, Interconnected, Interoperable
Customer Stories Written with Qt
"The customer experience, regardless of the mobile platform or OS, is the same by utilizing Qt."
Oncosoft uses Qt to boost real-time 3D medical imaging and more than double development efficiency.
-1.webp)
With Qt, Dräger were able to deploy their state-of-the-art UIs on resource-constrained hardware without compromising the performance.
.webp)
Qt equips Fresenius Kabi with efficient tools for designing medical devices and automating extensive testing to set new industry standards.

“Neocis was looking for a system that could perform on different operating systems for ease of use, and we found just that in Qt.”
.webp)
"To enhance efficiency, we modernized our development environment by adopting Qt for Mynatouch UI/UX development and unifying our programming languages."
MedTech Insights: Transforming Medical Device Software Development
From Conservative to Bold:
How MedTech Is Transforming Itself
This vision paper explores the drivers of modern MedTech innovation and outlines how a strong, reliable software foundation equips leaders to innovate without compromising regulatory integrity.
Safe and Effective Medical Device Development with Qt
Explore how Qt can advance your medical device development, ensuring safe, effective, and innovative solutions for your end-users. Packed with valuable insights and best practices that help you stay ahead of the competition.
Qt for MedTech Pre- and Post-Market Surveillance
Post-Market Surveillance (PMS) is a systematic process of monitoring and collecting data on the safety, performance, and effectiveness of a medical device after it has been placed on the market or put into use.
More About Medical UX, UI Design and Medical Software Development
Why MedTech Can No Longer Afford Poor UX/UI
Poor medical UI design can lead to serious risks in patient care. Intuitive UX design is thus no longer optional in medtech, also for its role in reducing costly errors and recalls.
Saving Lives with UX Design
Under high pressure, hospitals require staff to perform efficiently and safely, which is why effective UX design in healthcare aims to reduce cognitive load and minimize risks.
Read ArticleUniting Usability, Safety, and Security in Medical Devices
Intuitive UX design is not just for ease of use, but for ensuring patient safety and device security. Medical software usability cannot be separated from safety and cybersecurity.
Revolutionizing Healthcare: The Future of Intuitive Medical Devices
In this webinar, the leading MedTech experts discuss the cutting-edge innovations driving medical device software development forward.
Watch On-DemandStrengthening Healthcare Against Cyberattacks
The risk of cyberattacks is greater than ever in healthcare technology. We examine the strategies needed to safeguard patient data and maintain the integrity of life-saving systems.
Acing MedTech with Regulatory Intelligence
How do forward-looking companies in healthcare technology navigate the evolving regulatory environment and accelerate innovative medical software development?
Build Your Next Medical Software with Qt
Transform your medical devices, clinical lab equipment, and healthcare IT systems for full operability from home and anywhere in complete security.
When health and well-being are at stake, reliable and intuitive user interfaces to ensure medical software's usability, safety, and security are non-negotiable. Qt provides efficient tooling for designing and implementing effective healthcare UI applications and the extensive, automated testing and post-market surveillance needed to meet the highest certification standards.
